Home
Center Research
About Our Research
Center Research
Clinical Research
[Stage 4 Pilot Project] Prospective Observational Study in Patients with Shoulder Pain

1. Introduction
- This prospective observational study was conducted as part of the Korean Medicine and Western Medicine Collaboration Pilot Project.
- The collected data will be used to analyze the clinical and cost-effectiveness of collaborative treatment.
- Participants will receive usual care and participate in the study by completing surveys three times over an 8-week period.
- Shoulder pain is one of the most common musculoskeletal disorders. The number of patients seeking care for shoulder pain under the National Health Insurance (NHI) increased by 7.0% (an annual average of 1.7%), from 2.26 million in 2018 to 2.43 million in 2022.
- Shoulder pain is one of the most frequent conditions in the Korean Medicine and Western Medicine collaboration project, ranking second after low back pain in terms of the number of visits to Korean medicine clinics.
2. Participants
- Adult patients aged 19 years or older.
- New outpatients at institutions participating in the Korean Medicine and Western Medicine Collaboration Pilot Project.
- Patients diagnosed with a primary condition related to shoulder pain, based on the KCD codes listed below.
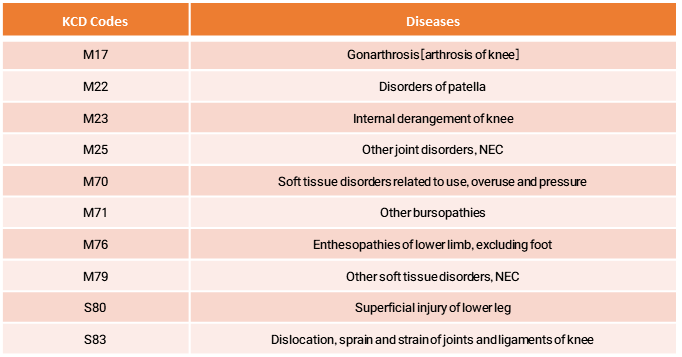
※ Exclusions : Patients covered by automobile insurance or participating in other research (except observational or survey studies).
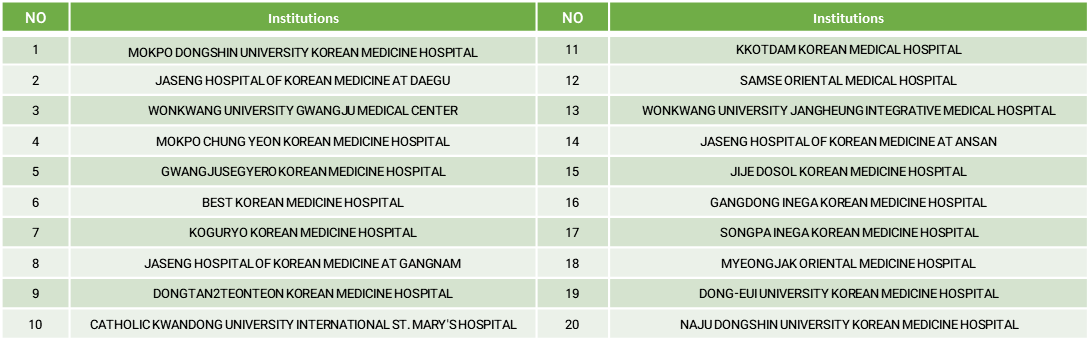
- List of participating medical institutions
3. Study Settings
1) Registration of Study Participants
- After the initial consultation, patients who are informed by the attending doctor and voluntarily agree to participate will be registered as study participants if they meet the criteria.
2) Medical Consultation
- The attending physician or Korean medicine practitioner will determine the need for collaboration and provide treatment. At this point, participants will be divided into those receiving collaborative treatment or those who do not require collaboration.
3) Survey Implementation and Data Collection
- The first survey will collect basic information from patients, including the severity of their condition and quality of life, conducted by the medical institution where the patient is receiving treatment.
- The second survey (4 weeks after the visit) and third survey (8 weeks after the visit) will be conducted by the Korean Medicine and Western Medicine Collaboration Monitoring Center via phone, collecting data on the progress of the condition, related quality of life, and medical expenses related to the condition (e.g., visits, hospitalization, surgery, treatments) over the past four weeks. The monitoring center staff will not know whether the patient is in the collaborative treatment group or the non-collaborative treatment group.
4) Result Analysis
- The collected data from the study participants and computerized data from the Health Insurance Review and Assessment Service (HIRA) will be analyzed to evaluate the clinical and cost-effectiveness of collaborative treatment.

4. Research Ethics and Safety
- This study will be conducted with approval from the Institutional Review Board (IRB).
- This is an observational study, and no risks are anticipated for the research participants.
- Information and data obtained from research participants will be protected and used in a reliable manner to ensure that their identities cannot be directly identified.
- Original data collected during the study will be stored for three years and then destroyed.
- Research participants have the right to withdraw their consent at any time.
- To ensure the fairness of the study, the Monitoring Center for Korean Medicine and Western Medicine Collaboration will be unaware of whether a research participant belongs to the collaboration group or the non-collaboration group during the second and third rounds of data collection.
- Old post
- There is no old post.